Chemical reactions in daily life
Chemical reactions can happen around us, not just in the laboratory. Material to build new products. Every time we cook or are clean, it is also an ingredient in action. Our bodies live and grow thanks to chemical reactions. There are reactions when we take medicine, spasms, and breathing. Here are 10 examples of chemical reactions in everyday life. This looks a small example, because we see and experience hundreds of thousands or even more chemical reactions every day.
1. Photosynthesis
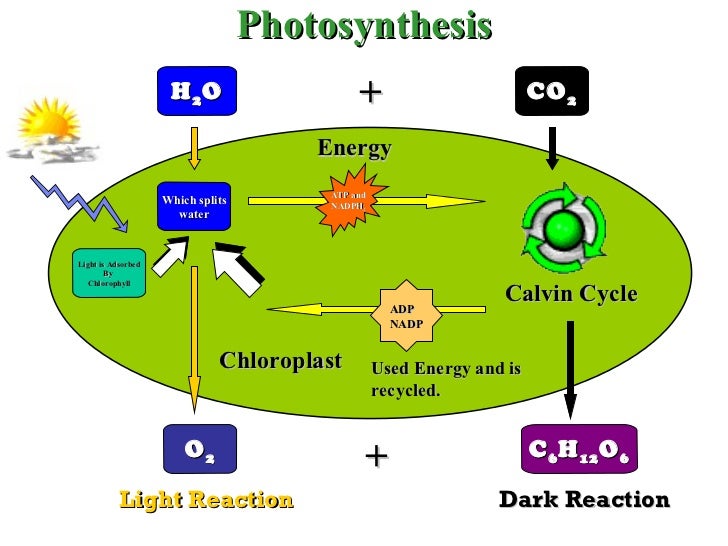





1. Photosynthesis

Photosynthesis is a
process used by plants and other organisms to convert light energy, usually
from the Sun, into a chemical energy that can be liberated to fuel organism
activity. This chemical energy is stored in carbohydrate molecules, such as
sugars, which are synthesized from carbon dioxide and water. In most cases,
oxygen is also produced as a waste product. Most plants, mostly algae, and
cyanobacteria do photosynthesis, and these organisms are called
photoautotrophs. Photosynthesis maintains atmospheric oxygen levels and
supplies all of Earth's organic compounds and most of the energy needed for
life on Earth.
Briefly,
plants use a chemical reaction called photosynthesis to convert carbon dioxide
and water into food (glucose) and oxygen. It is one of the most common
chemicals and reactions of carbon dioxide into oxygen.
=> 6 CO2
+ 6 H2O + light → C6H12O6 + 6 O2
2.
Burning

Every time we
light a match, burn a candle, make a fire, or light a grill, we will see a
burning reaction. Combustion combines energetic molecules with oxygen to
produce carbon dioxide and water.
For
example, propane combustion reactions, found in gas grills and some fireplaces,
are:
=> C3H8 +
5O2 → 4H2O + 3CO2 + energy
3. Rust

Rust is iron oxide, usually a red oxide formed by redox reactions of iron and oxygen in the presence of water or air humidity. Some forms of rust are distinguished both visually and by spectroscopy, and form under different circumstances. Rust consists of hydrated iron (III) oxide Fe2O3 · nH2O and iron (III) oxides-hydroxides (FeO (OH), Fe (OH) 3).
In sufficient time, oxygen, and water, every iron mass will eventually convert entirely of rust and crumbling. The rust surface is flaked and brittle, and provides no protection to the base iron, such as the formation of patina on the surface of the copper. Rust is a general term for corrosion of iron and its alloys, such as steel. Many other metals have equal corrosion, but the resulting oxide is not commonly called rust.
Here is the chemical equation for iron rust::
=> Fe + O2 + H2O → Fe2O3. XH2O
4. Battery

Batteries are electrical-chemical devices that store energy and emit energy in the form of electricity. A battery usually consists of three important components, namely:
1. carbon rod as anode (positive pole of battery)
2. zinc (Zn) as cathode (negative pole of battery)
3. pasta as electrolyte (conductor)
Batteries use electrochemical or redox reactions to convert chemical energy into electrical energy. Spontaneous redox reactions occur in galvanic cells, while chemical reactions do not occur spontaneously in electrolytic cells.
5. Digestion

Thousands of chemical reactions occur during digestion. As soon as we put food in the mouth, the enzyme in the saliva called amylase will begin to break down sugars and carbohydrates into a simpler form so our body can absorb it. The acid chloride in our stomach also reacts with food to break it down, whereas enzymes divide proteins and fats so they can be absorbed into the bloodstream through the intestinal wall.
6. Soaps and Detergents
Soaps and detergents can be cleaned by using chemical reactions. Soap emulsifies dirt, which means soap binds stain oil stains so they can be cleaned with water. The detergent acts as a surfactant, lowering the surface tension of the water so it can interact with oil, isolate them, and and clean it.



Please explain again the intent of point no 3 pasta as electrolyte (conductor) inside Battery
BalasHapusAmong the outermost layer of zinc acts as a negative pole and a mixture of manganese dioxide there is an ammonium chloride paste that serves as an electrolyte. Among the positive and negative poles there is a potential difference. It is this potential difference that causes the battery to carry an electric current when properly coupled in a circuit.
HapusPlease explain What causes rust?
BalasHapusCorrosion occurs because of the reaction between the metal and the substances around it. In rust, rust can occur because the humid air (oxygen and water) corrode (reacts) to the iron so that a new substance appears solid reddish brown
HapusAre there any harmful chemicals on detergent?
BalasHapusChemicals in detergents and detergents are very dangerous quartz ammonium groups. These compounds can form nitrosamine compounds known as toxins (carcinogenic) that can cause skin cancer.
Hapuswhat reaction happens to thr ?
BalasHapusit posible or not, alluminium can be rusting?
BalasHapusActually aluminum is also rusty, but can not be as porous as in iron. But, in extreme conditions aluminum can also be rusty and porous as in iron.
HapusPhotosynthesis maintains the level of oxygen in the atmosphere and supplies all the earth's organic compounds, mention examples of such organic compounds ?
BalasHapusProtein
HapusProteins are molecules made of amino acid monomers that are connected simultaneously
Carbohydrate
Carbohydrates are the long chain of sugar molecules that become one
Lipid
Lipids are molecules that form like oils so that lipids can be called fat
Nucleic Acid
Nucleic acid is a complex, high molecular-weight, biochemical macromolecule, and is composed of nucleotide chains containing genetic information.
Fat
Fat is a hydrophobic organic substance that is difficult to dissolve in water.
When the fire color in the combustion reaction is different, is the chemical reaction also different?
BalasHapusI think the color change proves the reaction, the reactant does not change but the reaction result will be formed. As an example As the sugar is heated continuously, the sugar changes color to black. The substance is carbon. Sugar turns into carbon and water due to heating.
HapusCarbon sugar + water
As a result of heating, a substance can change in two ways: experiencing compounding and decomposition. Compounding occurs as two or more substances join together to form a new substance, while decomposition is a chemical change due to a biodegradable substance into two or more new substances.